We know of many different antibiotics. And each of them kills bacteria through different mechanisms, attacking a specific vulnerability, their biological machineries.
So, when we take antibiotics because of a bacterial infection, billions of bacteria are suddenly attacked by antibiotics.
They struggle to repair damage, maintain their structures and continue essential functions just to stay alive.
This ultimately weakens or kills the cells.
And as you can imagine, this is pure stress for the bacteria.
One that we take advantage of.
This article series takes you on a journey through the microscopic war between bacteria and antibiotics. Across five articles, we will explore how antibiotics attack bacteria, how bacteria overcome them, become resistant and how evolution pushes bacteria to survive the antibiotic war.
In this first part of the series, we explore how different classes of antibiotics work, while focusing on the most commonly used antibiotics. Once you understand these mechanisms, you will better understand how and why bacteria fight back, evolve and develop resistance.
Antibiotics attacking bacterial cells by stopping cell division
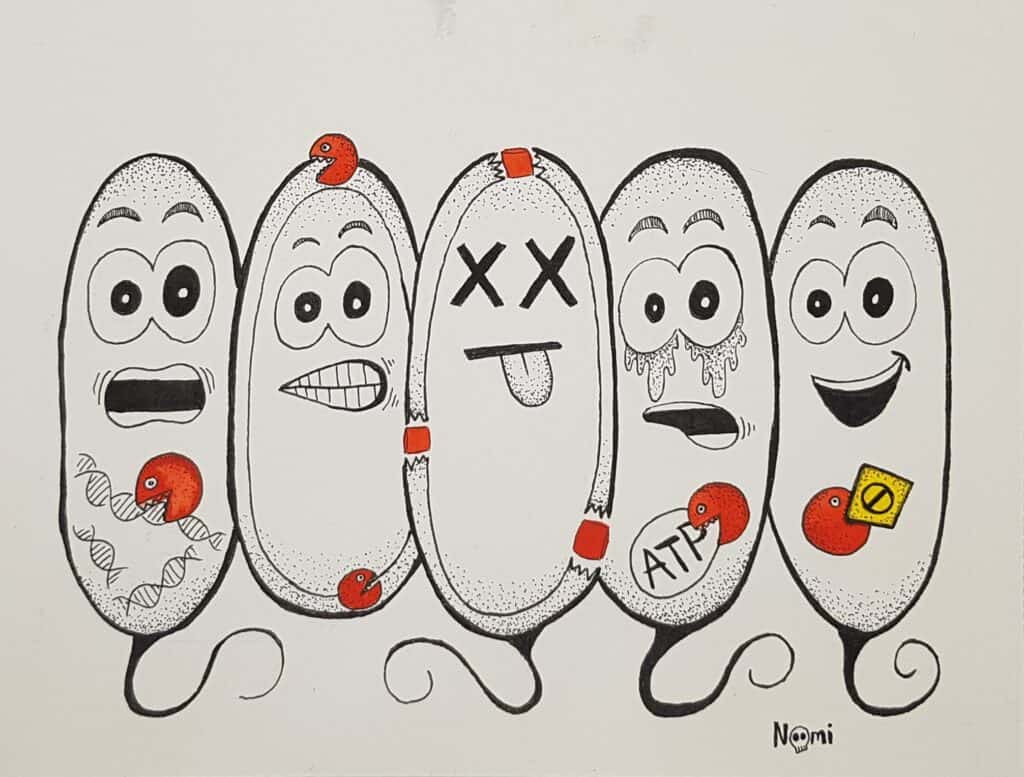
Bacteria have a rigid cell wall made of peptidoglycan to maintain their shape and internal pressure and to protect them from the environment. Without the ability to build or repair the cell wall, bacteria become fragile and burst easily.
This vulnerability is precisely what antibiotics from the β-lactam family exploit. You have probably heard of penicillin, one of the most well-known members of this class. Other similar antibiotics are amoxicillin and cephalosporins.
How these antibiotics work is pretty simple but devastating to bacterial cells: they block the so-called penicillin-binding proteins. These enzymes sit in the cell wall where they are responsible for building and cross-linking it.
So, when a bacterium gets hit by a β-lactam antibiotic, it loses the ability to divide. Basically, every time it tries to divide, it will burst like a water balloon.
Antibiotics sabotaging bacteria’s genetic machinery
Every bacterial cell carries instructions for life in its DNA, the molecule that stores genetic information. Before dividing, bacteria copy their DNA and then share it with their daughter cells.
Bacteria also make RNA, the molecule that executes the instructions stored in DNA. RNA comes in different types with distinct roles, but it is fundamentally needed to make proteins from DNA.
Some antibiotics exploit this vulnerability by inhibiting one of the cell’s information-processing machineries. This fundamentally interferes with DNA or RNA synthesis. If a bacterium can’t produce DNA or RNA, it can’t divide or maintain its genetic integrity, leading to cell death.
The antibiotic class fluoroquinolones inhibits DNA production. For example, ciprofloxacin freezes the enzymes that help bacteria copy their DNA. When DNA replication stalls, bacteria cannot divide. They accumulate damage and eventually die.
In comparison, rifamycins inhibit RNA synthesis. These antibiotics bind to the RNA polymerase, the enzyme that produces RNA from DNA. They thereby block the first step in protein production.
It’s like cutting electricity to an entire factory; without RNA, the cell cannot produce proteins, halting metabolism and growth. This is highly stressful to bacteria and can quickly kill them.
Antibiotics blocking protein production: The ribosome hijackers
Other antibiotics directly inhibit the protein production step: To build proteins, bacteria produce a temporary working copy of those DNA instructions, the so-called messenger RNA or mRNA.
This molecule travels to the ribosome, which reads the mRNA and makes proteins from it. Since proteins are essential for metabolism, movement, growth and cell division, no cell can function without them.
Some antibiotics, like tetracycline, take advantage of this protein production vulnerability. By interfering with ribosomes, these antibiotics prevent them from producing proteins and eventually the bacterial cells from functioning.
Interestingly, not all protein-production inhibitors kill bacteria in the same way. Some antibiotics are bacteriostatic, which means they freeze growth without immediately killing the cell. The bacteria cannot make new proteins, so they can’t divide or repair themselves. Instead, the existing proteins remain active for a while, allowing the cell to survive in a weakened state.
Others, like aminoglycosides, are bactericidal. Instead of simply blocking ribosomes, they cause the ribosome to make mistakes and produce misfolded proteins. These faulty proteins build up inside the cell and damage essential structures, overwhelming the bacterium, so it eventually dies.
Antibiotics disrupting metabolic pathways
Lastly, some antibiotics target essential metabolic pathways that bacteria need to survive. For example, folate is an essential vitamin that all organisms need to grow and reproduce, and most bacteria have proteins to make their own folate. So, when antibiotics block these folate-producing proteins, the bacterium will eventually run out of folate and lose the ability to grow.
How different antibiotics kill bacteria
As we’ve seen in this post, antibiotics can impact bacteria in many different ways. But they all have the same goal: do the biggest damage possible. Antibiotics can damage a bacterium’s DNA, its protein production machinery, metabolic pathways or the cell envelope.
This damage is essentially stress for the bacterium: they must repair the damage or adapt their metabolisms to it. If they cannot cope with the damage or the stress, they’ll die. And remember, this was basically the antibiotic’s goal from the beginning.
But be aware: this stress can be both lethal and a driving force for bacterial evolution. As they learn to cope with the antibiotic and the stress, they become resistant. And in future articles, we will explore these bacterial learning processes and how they help make bacteria resistant to antibiotics.